Gout
Last updated: November 25, 2014
ICD-9 Codes: Gouty arthritis, 274.0; gouty nephropathy, 274.1; uric acid nephrolithiasis, 274.11.
ICD-10: M10
Definition: Gout is an inflammatory disorder caused by tissue deposition of monosodium urate (MSU) crystals. Several descriptive terms are often used in association with gout:
—Acute gout: Single or recurrent attacks of inflammatory mono- or oligoarthritis.
—Tophaceous gout: Accumulation of crystalline MSU aggregates in soft tissues; nodular aggregates are referred to as tophi. Usually associated with chronic arthropathy.
—Hyperuricemia: Serum level of uric acid above which supersaturation of MS in extracellular fluids theoretically occurs (>6.8 mg/dL).
—Asymptomatic hyperuricemia: The state in which the serum uric acid level is abnormally high (>7.0 mg/dL in men, >6.0 mg/dL in postmenopausal women), but no symptoms of gout or nephrolithiasis have occurred.
—Primary gout: Gout resulting from abnormalities in purine metabolism or from idiopathic decreased renal excretion of urate.
—Secondary gout: Gout resulting from increased serum uric acid levels resulting from an associated disorder (e.g., neoplasms, lymphoproliferative disease, chronic renal failure) or drug therapy (e.g., diuretics, ethanol, cytotoxics).
—Saturnine gout: Gout caused by chronic lead intoxication, from either occult or occupational exposure or the ingestion of moonshine. This accounts for <5% of cases. Hyperuricemia results from lead-induced tubulointerstitial renal damage. Saturnine gout should be expected when the magnitude of hyperuricemia exceeds the reduction in glomerular filtration.
Etiology: The common denominator of gout is hyperuricemia. Uric acid, the product of purine degradation, is synthesized mainly in the liver. Two-thirds of the uric acid pool is excreted by the kidney, with the remainder secreted into the intestine. The causes of hyperuricemia can be divided into disorders of overproduction and disorders of decreased renal clearance of urate (Table 14). Most cases of gout (90%) are owing to underexcretion of uric acid; overproduction because of inherited enzyme defects [hypoxanthine-guanine adenine phosphoribosyltransferase deficiency (also known as Lesch-Nyhan syndrome) or overactivity of 5-phosphoribosyl 1-pyrophosphate synthetase] accounts for <1% of cases.
The pathogenesis of acute gout involves the response of polymorphonuclear leukocytes to formation of MSU crystals. Acute gout is thought to result from formation of new crystals rather than from release of crystals from preformed MSU synovial deposits or tophi. Crystals are coated with IgG, which reacts with Fc receptors on polymorphonuclear cells that phagocytose the crystals. Intracellularly, the crystals are stripped of their protein coats and disrupt the cell, releasing a variety of inflammatory mediators.
Pathology: The most frequent sites of MSU deposition are cartilage, epiphyseal bone, periarticular structures, and the kidney. Deposition in other sites is rare. Crystal aggregates cause a foreign body reaction. A tophus is composed of MSU crystals, a proteoglycan-rich intercrystalline matrix, and surrounding fibrous tissue. Affected joints may develop cartilage degeneration, erosion of marginal bone, and synovial proliferation. In the kidney, crystal deposition causes arteriosclerosis and interstitial fibrosis.
| Table 14: Causes of Hyperuricemia |
|---|
| Overproducers: increased purine synthesis or urate production |
| Idiopathic |
| Inherited enzyme defects |
| Hypoxanthine-guanine phosphoribosyltransferase deficiency |
| Complete (Lesch-Nyhan syndrome) |
| Incomplete |
| Phosphoribosylpyrophosphate synthetase overactivity |
| Diseases with purine overproduction |
| Lympho-andmyeloproliferativedisorders |
| Hemolyticdisorders |
| Malignant diseases |
| Obesity |
| Drugs and diet |
| Ethanol |
| Cytotoxic drugs |
| Warfarin |
| Purine-rich diets |
| Underexcreters: decreased renal clearance of urate |
| Primary idiopathic uric and underexcretion |
| Secondary uric acid underexcretion |
| Chronic renal failure |
| Hypertension |
| Dehydration |
| Obesity |
| Hyperparathyroidism |
| Hypothroidism |
| Drugs |
| Ethanol |
| Diuretics |
| Low-dose salicylate |
| Cyclosporine |
| Ethambutol |
| Pyrazinamide |
| Levodopa |
Demographics: The prevalence of asymptomatic hyperuricemia in adult Americans is 5% to 8%. The prevalence of gout is estimated to be 13 cases per 1,000 men and 6.4 cases per 1,000 women in North America. The risk of developing gout increases with higher uric acid levels: The annual incidence in relation to serum uric acid concentration is 0.1% for those with a serum uric acid level 9.0 mg/dL. More than 90% of patients with primary gout are men. Women rarely develop the disorder before menopause because estrogen is thought to enhance uric acid excretion. The peak incidence for men is in the fifth decade.
Risk Factors: Factors provoking episodes of acute gouty arthritis include trauma, surgery, immobility, alcohol ingestion, starvation, overindulgence in purine-rich foods, and drugs that raise urate concentrations (Table 14). Alcohol (ethanol) increases serum lactate levels, which blocks renal excretion of urate. Primary gout is often associated with obesity, hyperlipidemia, diabetes mellitus, hypertension, and atherosclerosis. Secondary gout may be associated with alcoholism and myeloproliferative and lymphoproliferative disorders. Gout is more prevalent among patients of African descent, largely because of the prevalence of hypertension.
Cardinal Findings
—Acute gout: Acute gouty arthritis is the most common early clinical manifestation of gout. The metatarsophalangeal joint of the first toe is the most common site (often referred to as podagra) of involvement, affecting 75% to 90% of patients at some time in the course of their disease, with 50% experiencing their first attack of acute gout in this joint. Approximately 80% of initial attacks are monarticular, typically involving joints of the distal lower extremity: the metatarsophalangeal joints, ankle, or knee. Wrist, finger, or elbow involvement is rare early but may occur with tophaceous gout or in the elderly with nodal OA, renal insufficiency, and diuretic use. Onset is usually abrupt, often waking the patient at night. The affected joints may be warm, swollen, and tender, with diffuse periarticular erythema that is often confused with cellulitis or thrombophlebitis. Fever may occur. Attacks may be associated with impressive soft tissue pitting edema. Early attacks generally subside spontaneously over 3 to 10 days, even in the absence of treatment. Postinflammatory desquamation of the skin overlying the joint may occur. Patients are typically symptom free after an acute attack. Subsequent episodes may occur more frequently, involve more joints, and persist longer. Trivial episodes of pain lasting a few hours may precede the first dramatic attack of acute gout. Although affected joints usually recover completely, erosions may develop in those with repeated attacks. Polyarticular attacks may also occur in those with established, poorly controlled disease. Such attacks may display a migratory or additive pattern or involve periarticular structures such as tendons and bursae.
—Intercritical gout: The interval between acute gouty attacks is termed the intercritical period. If necessary, MSU crystals can be recovered from previously affected joints in this symptom-free period. The duration of this period varies; most untreated patients experience a second episode within 2 years. A minority evolve into chronic polyarticular gout without pain-free intercritical periods. At this stage, the clinical picture can be confused with RA, especially if tophi are mistaken for rheumatoid nodules. However the two (RA, gout) rarely coexist because there is a negative association between the two.
—Chronic tophaceous gout: This form of gout is characterized by the deposition of solid urate (tophi) in connective tissues, including articular structures, with an eventual destructive arthropathy. Tophaceous gout may be associated with early age at onset, long duration of active untreated disease, frequent attacks, high serum urate levels, upper extremity involvement, and polyarticular disease. Organ transplant recipients treated with cyclosporine or diuretics are also at increased risk of accelerated development of chronic tophaceous gout. Common sites for tophi include the olecranon, prepatellar bursae, ulnar surface of the forearm, Achilles tendons, over the fingers, and the helix of the ear. Large tophi over the hands may occur in association with crippling joint destruction. Tophi may ulcerate and exude a white chalky material composed of MSU crystals. Tophi typically progress insidiously, with the patient reporting increasing stiffness and pain in affected joints.
—Renal disease: Renal disease includes urolithiasis, urate nephropathy (deposition of MSU crystals in the interstitium), and uric acid nephropathy (deposition of MSU crystals in the collecting tubules). Uric acid stones account for 5% to 10% of all renal stones. The prevalence of urolithiasis is 22% in primary gout and 42% in secondary gout, and renal stones antedate arthritis in 40% of cases. Serum uric acid levels are directly related to the incidence of urolithiasis (found in nearly 50% of patients excreting >1,100 mg of uric acid daily). Urate nephropathy and uric acid nephropathy are difficult to differentiate clinically, and they are often referred to as gouty kidney. Uric acid nephropathy may present acutely in patients with malignancy treated with chemotherapy or radiation. Urate nephropathy is slowly progressive and associated with hypertension and proteinuria. A causal relationship between renal dysfunction in gout and hypertension is equivocal.
Uncommon Findings: Uncommon sites of initial involvement are the hands, shoulders, sternoclavicular joints, hips, spine, and sacroiliac joints. Initial presentation of gout may be polyarticular, especially in women. Aseptic necrosis of the hip has been reported as a manifestation of gout. Tophaceous involvement of the axial skeleton has been rarely noted. Tophaceous involvement of parenchymal organs, although rare, has been also been reported. Uncommonly, atypical gout arises in nodal OA with acute inflammatory swelling affecting the Heberden or Bouchard nodes. Such patients tend to be elderly females with renal insufficiency, often treated with diuretics.
Diagnostic Tests: At initial presentation, serum uric acid levels are elevated, although levels are normal in as many as 40% of patients experiencing an acute gouty attack. Nonetheless, the vast majority of patients with gout demonstrate an elevated uric acid level at some time. The serum creatinine level should be determined because it may influence subsequent therapy. During an acute attack, there is frequently leukocytosis, thrombocytosis, and elevated acutephase reactant (ESR, CRP) levels.
—Joint fluid: Synovial fluid in acute gout is inflammatory, with high leukocyte counts (>2,000 cells/mm3; WBC differential >75% neutrophils); occasionally, synovial leukocyte counts are very high (>50,000 cells/mm3). MSU crystals are identified with the polarized light microscopy. Needle-shaped crystals, 5 to 25 J..m, can be seen against a dark background. These are best identified with polarizing lenses and a red compensator, which reveals characteristic negatively birefringent crystals that appear yellow against a lilac background when parallel to the plane of the red compensator and blue when at right angles to it. In acute gout, urate crystals are usually intracellular (within neutrophils); between attacks, urate crystals may still be seen, but they tend to be extracellular. Only one or two drops of synovial fluid are necessary for crystal examination. Needle-shaped MSU crystals can also be seen under plain light microscopy. Demonstration of MSU crystals does not exclude pseudogout or septic arthritis because these conditions may coexist with gout. If septic arthritis is considered, Gram staining and culture should be performed. Exudate from gouty tophi can be examined for MSU crystals in a similar manner.
—Urine tests: A 24-hour collection for uric acid determination is useful for assessing the risk of renal stones and for planning therapy (if use of a uricosuric agent is contemplated).
Imaging: Radiographic examination in acute gout is most valuable in excluding other types of arthritis (e.g., septic arthritis, fracture). Radiographic abnormalities seen in longstanding gout are an asymmetric, erosive arthritis with characteristic “scooped-out” marginal erosions with sclerotic borders and often an overhanging edge of cortical bone. Periarticular osteopenia is absent, and the joint space is preserved until late in the disease.
Keys to Diagnosis: A history of podagra, dramatic onset of arthritis, history of episodic or prior arthritis with spontaneous resolution in 3 to 10 days, and the presence of tophi should all suggest a diagnosis of gout. For the diagnosis of acute gout, serum uric acid concentrations are neither sensitive (often not raised in acute attacks) nor specific. Definitive diagnosis rests on demonstration of MSU crystals within leukocytes in affected joints. A scoopedout marginal joint erosion with sclerotic borders and an overhanging edge also suggests gout.
Differential Diagnosis: The differential diagnosis depends on the stage of gout encountered.
— Acute gout: The differential diagnosis of acute gout is essentially that of acute inflammatory monarthritis and includes septic arthritis, reactive arthritis, ARF, pseudogout, and other crystalline arthropathies. Fever, leukocytosis, and localized erythema may lead to erroneous consideration of cellulitis or thrombophlebitis (when the distal lower extremity is involved).
— Chronic tophaceous gout: The differential diagnosis of chronic tophaceous gout includes other destructive arthropathies, RA, chronic CPPD crystal deposition disease, seronegative SpAs, and erosive OA. Tophi are often mistaken for rheumatoid nodules because of their appearance and location. Coexistence of gout and RA is exceedingly rare.
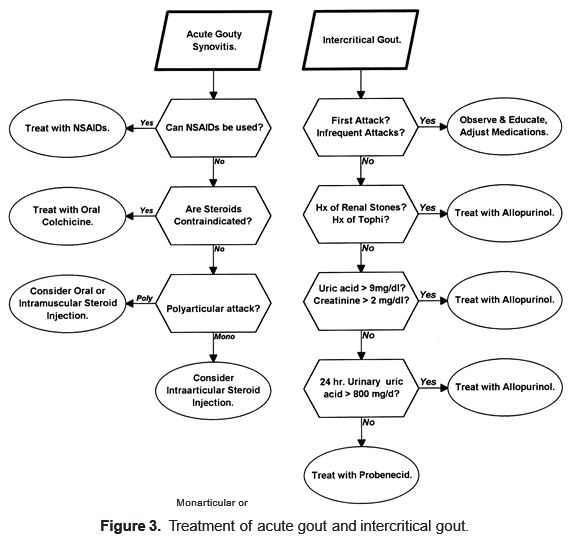
Therapy: The goal of therapy is to treat the acute attack of gout and prevent recurrent attacks and the complications of untreated gout. There is usually no justification for treating asymptomatic hyperuricemia. However, some physicians make exceptions when serum urate levels are very high (e.g., >12 mg/dL) and the risk of nephrolithiasis is substantial. In addition, allopurinol therapy may be indicated in the setting of malignancy, when the anticipation of tumor lysis results in acute overproduction of urate. Treatment options vary with the stage of gout encountered (Fig. 3).
—Acute gout: NSAIDs, corticosteroids (locally or systemically), or oral colchicine may be used (Fig. 3). NSAIDs are the preferred modality in acute gout because they have a rapid clinical effect and are usually well tolerated when used for 3 to 14 days or until the attack subsides. Nonetheless, NSAIDs may be associated with acute gastritis and renal toxicity and should be avoided in those with a history of GI intolerance, renal insufficiency, congestive heart failure, ascites, bleeding diathesis, or chronic anticoagulant therapy. Although indomethacin (1–2 mg/kg/day) is historically the most widely used NSAID for treatment of acute gout, other nonselective NSAIDs and COX-2 inhibitors have been effective at antiinflammatory doses. When NSAIDs are contraindicated, corticosteroids are usually effective. They may be particularly useful in the elderly, in persons with renal insufficiency or congestive heart failure, and in organ transplant recipients—all situations in which NSAIDs and colchicine may be relatively contraindicated. Intraarticular preparations (e.g., methylprednisolone acetate) are particularly efficacious in monarticular gout. Oral or parenteral steroids are also effective in the treatment of monarticular and polyarticular gout. Historically, colchicine therapy has been used to control acute attacks. However, unacceptable GI toxicity (i.e., nausea, diarrhea) and delayed onset of action make this the least option in acute gout. In patients with normal renal function, colchicine can be administered orally with a dose of 1.2 mg initially, followed by 0.6 mg every 2 hours, until abdominal discomfort or diarrhea develops or a total dose of 8 mg has been administered. Most patients have some relief of arthritic symptoms by 18 hours, with joint inflammation subsiding in 48 hours. Colchicine’s effect relates to the inhibition of microtubule formation and neutrophil chemotaxis. Intravenous colchicine should not be used (see Comments below). Antihyperuricemic measures (i.e., allopurinol) should not be initiated or continued until the acute attack is resolved because the duration of joint inflammation may be prolonged. Doses of allopurinol and colchicine must be adjusted with renal insufficiency.
—Intercritical gout: Once the acute attack has resolved, attention is directed at prevention and prophylaxis. The decision to initiate chronic pharmacotherapy is made in accord with the patient’s wishes (Fig. 3). Those with one or few attacks may prefer to wait and treat the attack, when and if it arises. Those with multiple attacks should be offered the opportunity to prevent attacks with medical therapy. Prevention can be achieved by correcting hyperuricemia, either by eliminating identifiable causes of hyperuricemia (diuretic therapy) or by administering drugs that lower uric acid production or enhance its excretion. Complete avoidance of purine-rich foods (meats, yeast, alcohol, legumes, spinach, asparagus, cauliflower, and mushrooms) is impractical, ineffective, and rarely adhered to in clinical practice. Gout can be prevented or diminished by lowering serum uric acid levels with uricosuric agents (probenecid, sulfinpyrazone) or by inhibiting production of uric acid (allopurinol). Indications for pharmacologic lowering of serum uric acid include the inability to reverse secondary causes of gout, recurrent attacks of gout arthritis, chronic tophaceous gout, and an increased risk of nephrolithiasis. In general, use of uricosuric agents is limited to persons with normal renal function, decreased urinary urate excretion (mg/day. Therapy may be unsuccessful in some patients. Allopurinol effectively lowers uric acid levels in both overproducers and underexcreters of urate and is indicated in persons with overproduction of uric acid, renal stones, tophi, renal insufficiency, and extreme elevation of uric acid and those in whom uricosuric agents are contraindicated or are ineffective. Allopurinol is also indicated in the prophylaxis of tumor lysis syndrome. The usual dose of 300 mg/day must be reduced in the presence of renal insufficiency (200 mg/day for a creatinine clearance <30 mL/min). Beginning with small doses reduces the likelihood of precipitating an attack of acute gout. The chief side effect of allopurinol is a rash that develops in as many as 2% of patients and can occasionally be life threatening if exfoliative dermatitis develops (one in 1,000 cases). Chronic colchicine therapy (0.6–1.2 mg/day) may also be prophylactic against gouty attacks. The dose must be adjusted for renal insufficiency. Neuromyopathy or rhabdomyolysis is a rare side effect strongly associated with renal insufficiency.
Prognosis: Untreated, gout progresses over several years from the initial attack of acute gout through a period of intercritical gout to chronic tophaceous gout. More rapid progression may be seen in the presence of severe renal insufficiency. Early intervention can interrupt this progression. Tophi can regress completely with prolonged treatment. Occasionally, gout is difficult to manage, often because of multiple factors such as compliance, alcoholism, renal insufficiency, and the need for continued diuretic therapy.
Comment: The clinician should refrain from using intravenous colchicine to treat acute gout because of the risk of acute bone marrow suppression and hepatic, renal, and CNS toxicity, especially in those with renal impairment. Although the intravenous preparation is available in the United States, it is banned in many formularies and in many other countries, including the United Kingdom and Australia.
BIBLIOGRAPHY
Boomershine KH. Colchicine-induced rhabdomyolysis. Ann Pharmacother 2002;36:824–826. Emmerson BT. The management of gout. N Engl J Med 1996;334:445–451.
Halla JT, Ball GV. Saturnine gout: a review of 42 patients. Semin Arthritis Rheum 1982;11:307–314.PMID:6821383
Kim KY, Ralph Schumacher H, Hunsche E, et al. A literature review of the epidemiology and treatment of acute gout. Clin Ther 2003;25:1593–1617. PMID:12860487
Rott KT, Agudelo CA. Gout. JAMA 2003;289:2857–2860.PMID:12783917